biogeochemical modelling as a key tool for oyster reef restoration success
Author: Tahlia Martignago
Date: 18 August 2023
Oysters are bivalve molluscs that are usually distributed across temperate and warm coastal and estuarine waters. When occurring in dense aggregations, they form reef structures comprised of both living organisms and dead shell accumulations. These biogenic structures act as the foundation of the ecosystem through their contribution to the habitat and protection of a wide variety of marine species as well as the provision of critical ecosystem services, including water filtration, nutrient cycling, erosion mitigation and pH buffering (Gillies et al., 2020).
Unfortunately, European settlement resulted in a significant reduction to Australia’s oyster reefs (McAfee et al., 2022). Prior to colonisation, reefs were a significant feature of Australia’s coastal and estuarine regions, covering over 7,000km. European settlers quickly exploited oyster reefs as a critical resource for the development of the early European colony, burning their calcium carbonate shells for the production of lime for cement manufacture (Luders, 2017). Today, there is only one small natural Flat Oyster (Ostrea angasi) reef and 6 remnant Sydney Rock Oyster (Saccostrea glomerata) reefs remaining across all of Australia, less than 1% of Australia’s historic reefs. With oyster reefs largely missing from our coastlines, the nutrients, sediments, and other runoff occurring as a result of urbanisation have greatly reduced our coastal water quality, and fish stocks and other marine life are in decline due to a lack of reef area to colonise and feed (The Nature Conservancy, n.d.).
As a result, increased efforts have now been introduced to restore Australia’s shellfish reefs, with many programs dedicated to the identification, restoration and protection of shellfish reefs across the country. However, monitoring associated with oyster reef restoration projects has been found to be inadequate, with “little post-construction monitoring to allow for comparison among restoration projects, adaptive management, and determination of whether restoration goals were successfully achieved” (Beggett et al., 2014). In the more recent restoration projects headed by The Nature Conservancy Australia, it has been noted that initial reef monitoring is only temporarily sustained, with “handover and closeout” procedures currently in progress with no mention of ongoing monitoring efforts (TNC Oceans Program, 2021).
Consistent monitoring and modelling of both environmental and biological variables would be highly beneficial to the ongoing success of restored reefs through informing adaptive management procedures (Pine et al., 2022). Climatic conditions and the subsequent availability of resources are critical influences on physiological and behavioural processes of reproduction, development and growth in many organisms, and thus have the potential to help or hinder restoration efforts (Kennedy et al., 1996). The growth performance of oysters has been found to depend on the availability of chlorophyll, amongst other environmental variables (Cugier et al., 2022). Chlorophyll a, as a proxy for net food contributions, significantly affects oyster growth and function, with diatoms constituting a significant food source (Mizuta et al., 2012; Kim et al., 2019). In addition, pH, as a significant influence on the bioavailability of calcium carbonate, also has the potential to contribute to restoration outcomes, as a lower pH decreases the availability of calcium carbonate, which are a critical resource in the formation, growth and maintenance of oyster shells (Lemasson et al., 2017).
Through this exploratory study we aimed to investigate how environmental variables impact oyster growth and mortality outcomes in order to develop insights as to how oyster restoration projects may use environmental modelling as a tool for success. Using a 26 year time series dataset relating to the mass and mortality of the Pacific Oyster (Crassostrea gigas) at 13 sites along the French coast, we were able to plot growth and mortality of juvenile, half grown oysters as a function of time. This data was compared to environmental conditions of the area, including dissolved oxygen, nitrate, phosphate, chlorophyll-a, phytoplankton and pH, derived from a global model. The Global Ocean Biogeochemistry Hindcast by the Copernicus Marine Service offers three-dimensional biogeochemical data covering the period from 1993 to 2019. This data is generated using the PISCES biogeochemical model, which is accessible through the NEMO modeling platform. The PISCES model has a spatial resolution of 0.25 degrees, so the data point that was nearest to or encapsulating the site of interest was used to observe how environmental data changed at a single location over time.
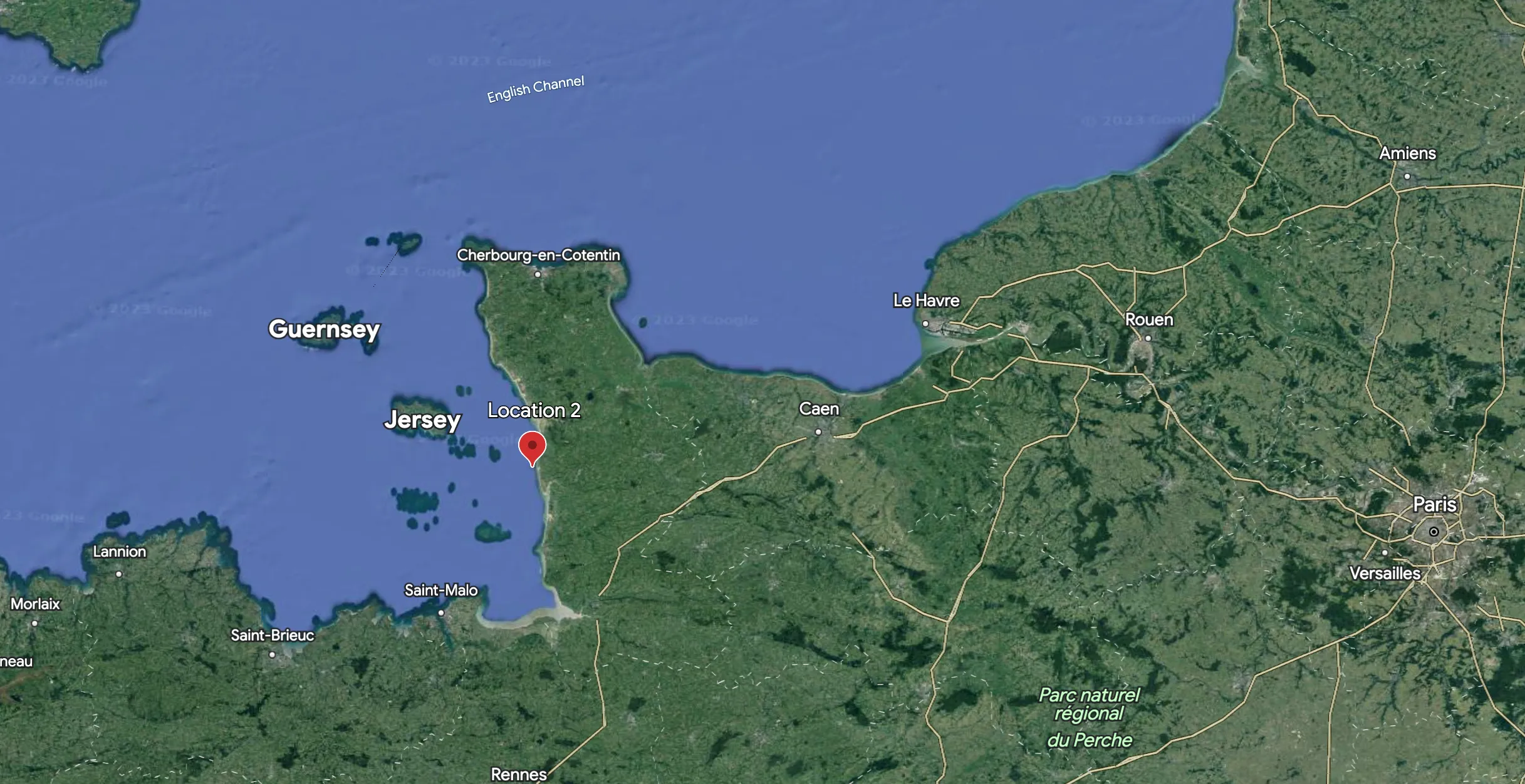
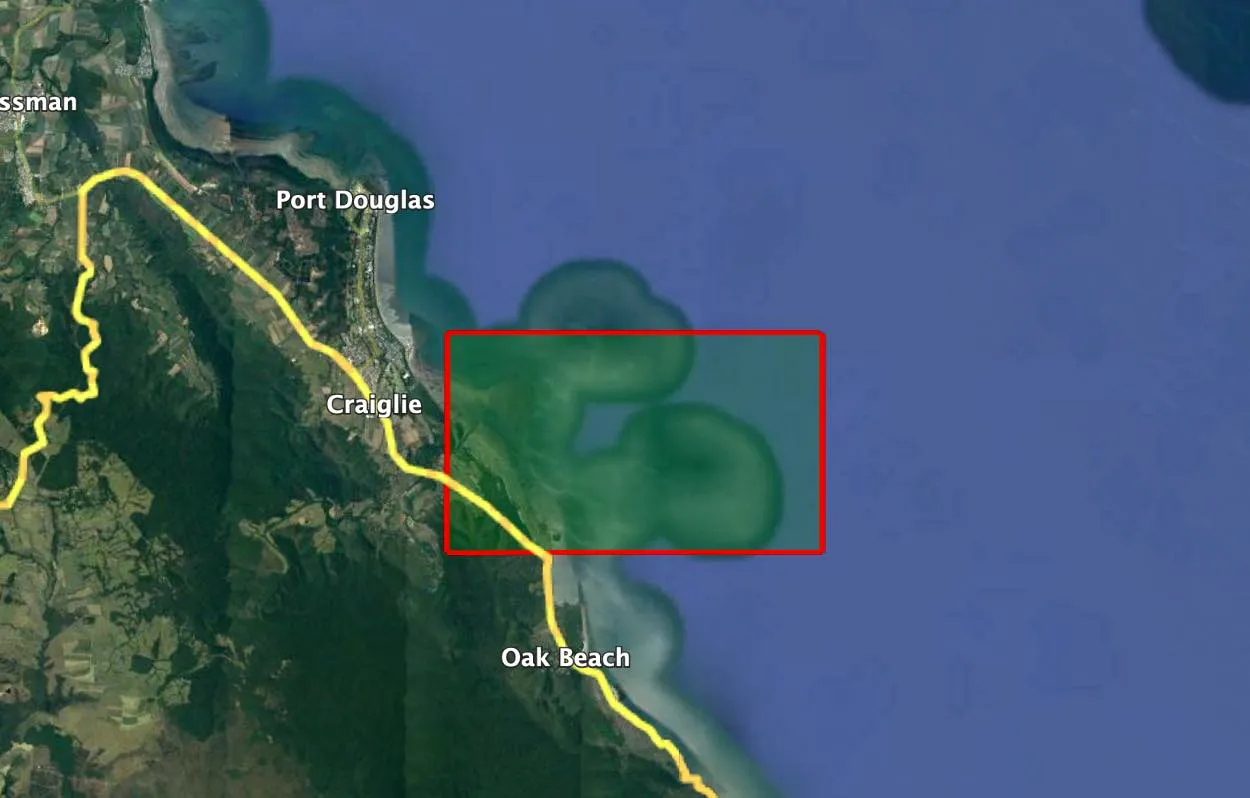
Figure 1: ‘Location 2' denotes the area of interest along the French coast (49.065780, -1.629950). This location is the second of 13 monitored sites that comprise the 26-year time series dataset (Source: Google Earth).
Oyster mortality did not appear visually correlated to any available biogeochemical data, where a considerable increase in oyster mortality during 2008–2009 was not accompanied by any noticeable change in biogeochemical parameters (Figure 2). This may be a result of the limited accuracy of the NEMO global model in a regional context, but may also suggest the presence of other external factors contributing to oyster mortality. This phenomenon was consistent amongst most sample locations and agrees with the abnormally high mortality rates in Pacific oysters observed along the French coast in the summer of 2008 that was found to be a result of an outbreak of an Ostreid herpesvirus-1 outbreak (OsHV-1) (Segarra et al., 2010). This suggests the drawbacks of purely using modelled biogeochemical data, and thus the need for diverse monitoring providers and goals to provide a comprehensive understanding of other variables such as pathogens, ecosystem biodiversity and interspecific competition that may contribute to decreased probabilities of successful restoration beyond water quality parameters.
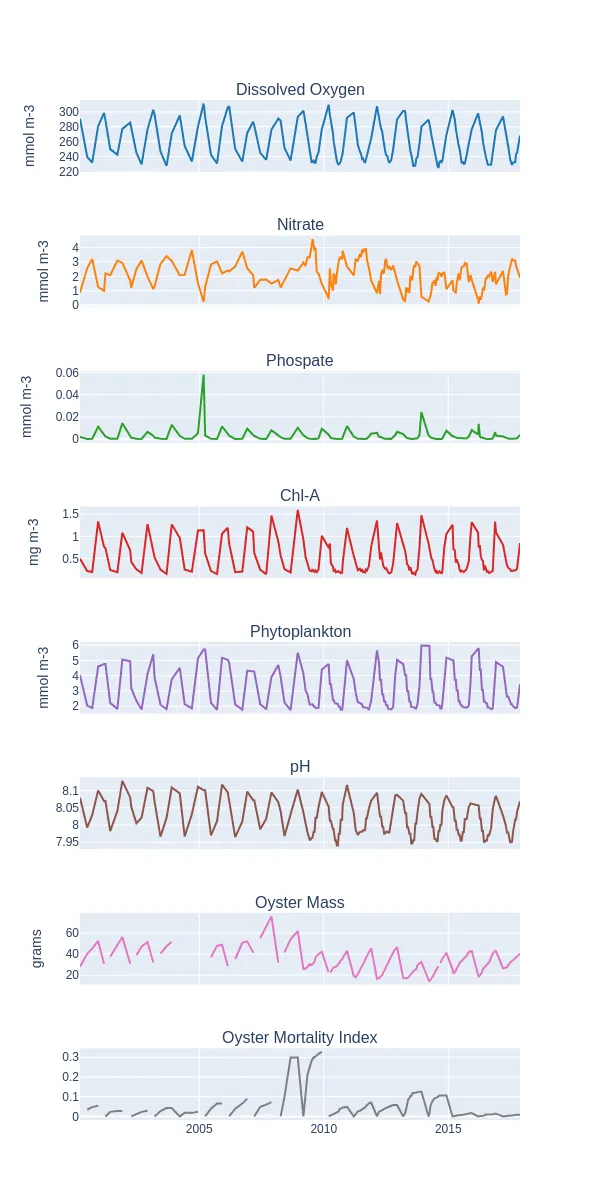
Figure 2: Biogeochemical data derived from the NEMO model as compared to the oyster mass and mortality data at Location 2 between 2000 and 2017. All parameters exhibit seasonality, with periodic peaks and troughs occurring during the summer and winter seasons. Oyster mortality showed a considerable increase between 2008–2009 which was preceded by a large increase in oyster mass.
Oyster mass was observed to spike in November 2007 prior to the increase in oyster mortality in September 2008 (Figure 3). Following this mortality event, oyster mass on average appears lower than before the event. This is consistent with a mass mortality event occurring in the Mississippi Sound, USA in 2016, whereby it was found that oyster weights did not return to levels observed before the event, and the size frequency of the population following the mortality event favoured smaller individuals (Pace et al., 2020).
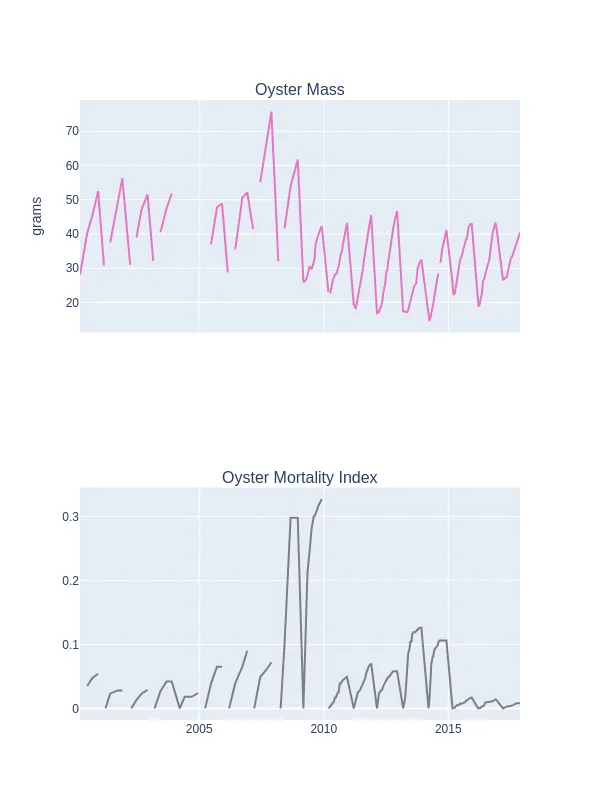
Figure 3: Oyster mass and oyster mortality between 2000 and 2017 at location 2 along the French coast (Mazaleyrat et al., 2022). Both oyster mass and mortality appear seasonal, however they considerably increased in November of 2007 and September of 2008, respectively.
Whilst statistical analyses are needed to draw precise conclusions or correlations, pH is observed to decrease throughout the measured time period of 2005 to 2015 (Figure 4). This may be associated with the overall decrease in oyster mass over time observed in figure 3. Since oyster mass measured both oyster tissue and shell (Mazaleyrat et al., 2022), this decrease in pH may contribute to a change in oyster shell composition and density, thereby reducing oyster mass (Meng et al., 2018). Decreases in pH have also been associated with negative outcomes in relation to shell planting as a main component of oyster restoration efforts, where dead oyster shell is planted to initiate oyster recruitment (Waldbusser et al., 2011).
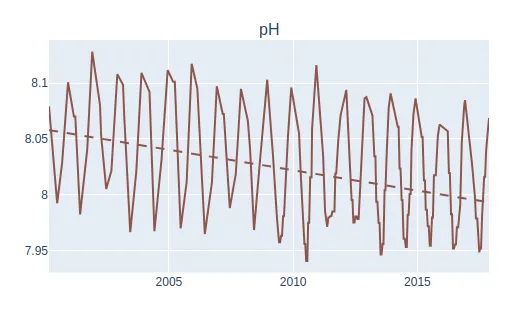
Figure 4: Despite seasonality, decreases in pH was observed at Location 2 and its surrounding areas between 2000 and 2017 (Mazaleyrat et al., 2022).
Reliance on biogeochemical models for the maintenance and protection of restored oyster reefs faces many limitations. At the moment, in situ measurements and real-time remote sensing data is predominantly used for oyster restoration projects. The NEMO model as used in this exploratory study is a global model, with limited data available for estuarine locations, the predominant environment for reef restoration projects in Australia, particularly New South Wales. The use of CSIRO’s regional model would be beneficial for further exploration of biogeochemical parameters within these Australian locations. Additionally, the NEMO model lacks fine resolution that would enable a more accurate and reliable representation of the biogeochemical properties at specific locations. However, this project was not intended as a rigorous scientific study, instead, we show that biogeochemical parameters can be derived from a global model to be used in conjunction with biological data as an important contributor to predicting the health and success of oyster restoration projects. This project was only an exploration of what could be obtained quickly with the NEMO model, and a tentative investigation into the potential correlations between environmental data and biological data derived from oyster restoration projects.
This study has been conducted using E.U. Copernicus Marine Service Information; https://doi.org/10.48670/moi-00019
About the authors —
Deepti Mallampalli and Tahlia Martignago are students at the University of Technology Sydney, where Deepti studies a Bachelor of Computer Science, majoring in Data Analytics and AI, whilst Tahlia studies a Bachelor of Science (Environmental Science) and a Bachelor of Creative Intelligence and Innovation. This project took place through the internship programs offered at UTS under the supervision and expertise of Dr Iwan Cornelius of Amentum Scientific. A big thank you is also extended to Thomas Pugh, for their expertise in developing the interface for the NEMO model that was crucial to obtaining the data we used in this study. We have been so grateful for the opportunity to receive first-hand industry experience, and have learnt so much about modelling as a tool for oceanic monitoring, and how to interpret this data with biological and industry relevance.